⚛️ Real Gas Equation Calculator
(P + a(n/V)2)(V − nb) = nRT
Table of Contents
✍️ Author & Academic Authority: Dr. Nitish Kr. Bharadwaj
📘 Qualifications: B.Sc., B.Ed., M.Sc., Ph.D. (Biochemistry), MBA (Financial Management)
🧪 Real Gas Equation Calculator – Introduction
Instantly Solve Real Gas Law Problems 🚀
Understanding how gases behave is one of the most important concepts in thermodynamics and physical chemistry. While many students learn about the Ideal Gas Law, real gases in nature do not always behave ideally. That’s where the Real Gas Equation, commonly known as the Van der Waals Equation, becomes essential.
The Real Gas Equation Calculator is a powerful online physics tool designed to help students, researchers, and professionals quickly compute gas properties such as pressure, volume, temperature, and moles when dealing with non-ideal gas behavior.
Unlike ideal gas calculations, which assume that gas molecules occupy no volume and experience no intermolecular forces, real gases behave differently under high pressure and low temperature conditions. The Van der Waals equation corrects these limitations by introducing constants that account for molecular attraction and finite molecular size.
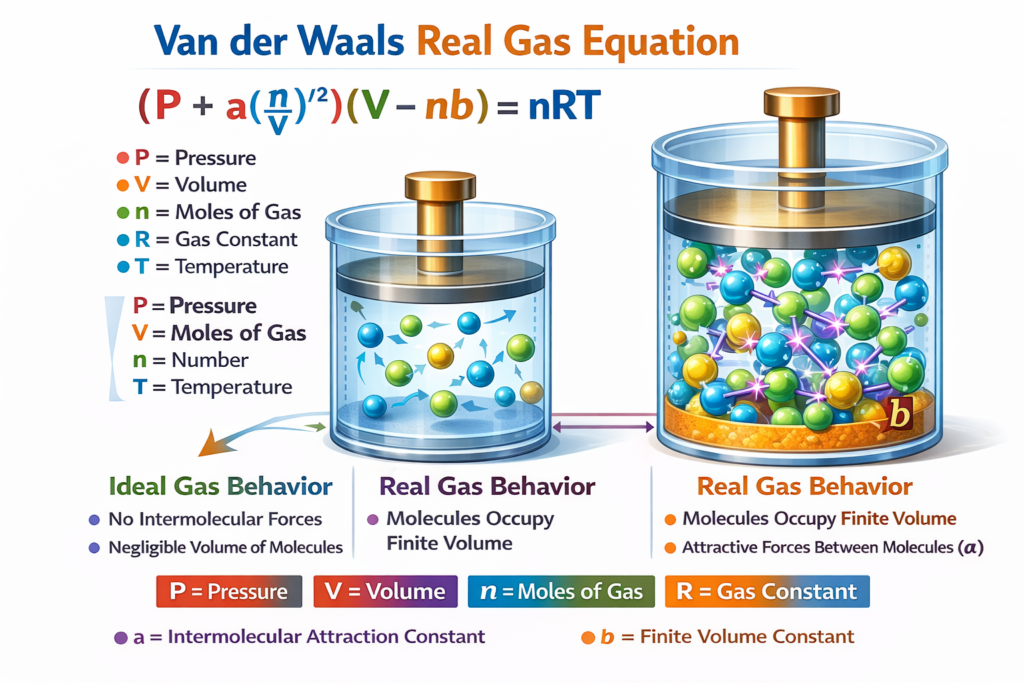
The general form of the Real Gas Equation is: (P + a(n/V)²)(V − nb) = nRT
Where:
P = Pressure of the gas
V = Volume of the gas
n = Number of moles
T = Temperature (Kelvin)
R = Universal gas constant
a = Van der Waals constant representing intermolecular attraction
b = Van der Waals constant representing molecular volume
Using the Real Gas Equation Calculator, users can simply input the required variables and instantly obtain accurate results without manual calculations.
🔬 Why Real Gas Calculations Matter
In real-world conditions, gases rarely behave perfectly like the Ideal Gas Law (PV = nRT) predicts. This is especially true when:
✔ Pressure is very high
✔ Temperature is very low
✔ Gas molecules interact strongly
Under these conditions, the real gas law equation becomes crucial for accurate predictions.
The Van der Waals equation calculator simplifies this process by performing complex thermodynamic calculations instantly.
⚛️ Key Features of the Real Gas Equation Calculator
✨ Instant Calculations: Solve real gas equation problems in seconds.
📊 Accurate Thermodynamic Results: Accounts for molecular attraction and volume corrections.
🎓 Perfect for Students & Educators: Useful for physics, chemistry, and engineering problems.
📱 Mobile Friendly Tool: Works on phones, tablets, and desktops.
⚡ No Manual Formula Application Needed: Just enter values and get results instantly.
🧠 Real Gas vs Ideal Gas Behavior
Understanding the difference between real gases and ideal gases is essential in thermodynamics.
Ideal Gas Assumptions:
- Gas molecules have no volume
- No intermolecular forces
- Collisions are perfectly elastic
Real Gas Reality:
- Molecules occupy finite space
- Intermolecular attractions exist
- Behavior changes at extreme conditions
Because of these differences, the Van der Waals equation corrects the ideal gas law by introducing constants a and b, making it more accurate for real-world gas systems.
🌍 Importance of Real Gas Equations in Science
The Real Gas Equation plays a vital role in multiple scientific and engineering applications including:
- Chemical reaction modeling
- Industrial gas storage
- Atmospheric science
- Thermodynamic system design
- Refrigeration systems
- Aerospace engineering
By accurately predicting gas behavior, scientists and engineers can design safer and more efficient systems.
📈 Why This Calculator is Helpful for Learning
Students often struggle with thermodynamics equations because they involve multiple variables and constants. The real gas equation calculator online makes learning interactive and intuitive.
By experimenting with different values, students can observe how pressure, temperature, and volume influence gas behavior, helping them build stronger conceptual understanding.
🌍 Applications in Daily Life
Although the real gas equation is commonly studied in physics and chemistry, it also has practical applications in everyday technologies.
🏭 Industrial Gas Storage: Gas cylinders storing oxygen, nitrogen, and carbon dioxide use real gas law calculations to determine safe pressure limits.
❄️ Refrigeration Systems: Refrigerators and air conditioners rely on real gas thermodynamic behavior for cooling cycles.
🚀 Aerospace Engineering: Rocket propulsion systems use real gas equations to analyze fuel behavior under extreme pressures.
🌡️ Weather & Atmospheric Science: Meteorologists use real gas models to understand atmospheric pressure and temperature variations.
⚗️ Chemical Engineering: Chemical plants apply Van der Waals equation calculations to design reactors and pipelines.
⚠️ Disclaimer
The Real Gas Equation Calculator provided on this website is intended for educational and informational purposes only. While every effort has been made to ensure the accuracy of the calculations, the results should not be used as the sole basis for engineering, industrial, or safety-critical decisions.
Users should verify results independently and consult qualified professionals when applying thermodynamic calculations in real-world applications.
By using this calculator, you acknowledge that allcalculators.co.in is not responsible for any errors, inaccuracies, or decisions made based on the results generated by this tool.
📌 Related Calculator
❓ FAQs
❓ What is the Real Gas Equation?
The Real Gas Equation, also known as the Van der Waals equation, is a thermodynamic formula that describes the behavior of real gases by accounting for intermolecular forces and molecular volume.
❓ What is the difference between ideal gas and real gas?
The Ideal Gas Law assumes gas molecules have no volume and no attraction between them, while real gases have intermolecular forces and occupy finite space, which the real gas equation corrects.
❓ When should the Real Gas Equation be used?
The real gas law should be used when gases are at high pressure or low temperature, where the ideal gas law becomes inaccurate.
❓ What are Van der Waals constants?
The Van der Waals constants (a and b) are gas-specific parameters that represent intermolecular attraction (a) and molecular volume (b).
❓ Why is the Real Gas Equation important in physics?
The real gas equation provides a more accurate model of gas behavior in real-world conditions, making it essential for thermodynamics, chemical engineering, and industrial applications.
