⚛️ Dipole Moment Calculator
Calculate Electric Dipole Moment using μ = q × d with proper scientific notation & unit conversion 🔬
μ = q × d
Where:
μ = Dipole Moment (C·m or Debye)
q = Charge (Coulomb)
d = Separation Distance (meter)
⚡ Dipole Moment Calculator – Understand Molecular Polarity and Charge Separation Easily 🧪
The Dipole Moment Calculator is a powerful physics and chemistry tool that helps students, educators, and researchers determine the dipole moment of a molecule or charge system quickly and accurately. Dipole moment is a fundamental concept in electrostatics, molecular chemistry, and quantum physics, and it plays a vital role in understanding molecular polarity, intermolecular forces, chemical bonding, and reaction behavior.
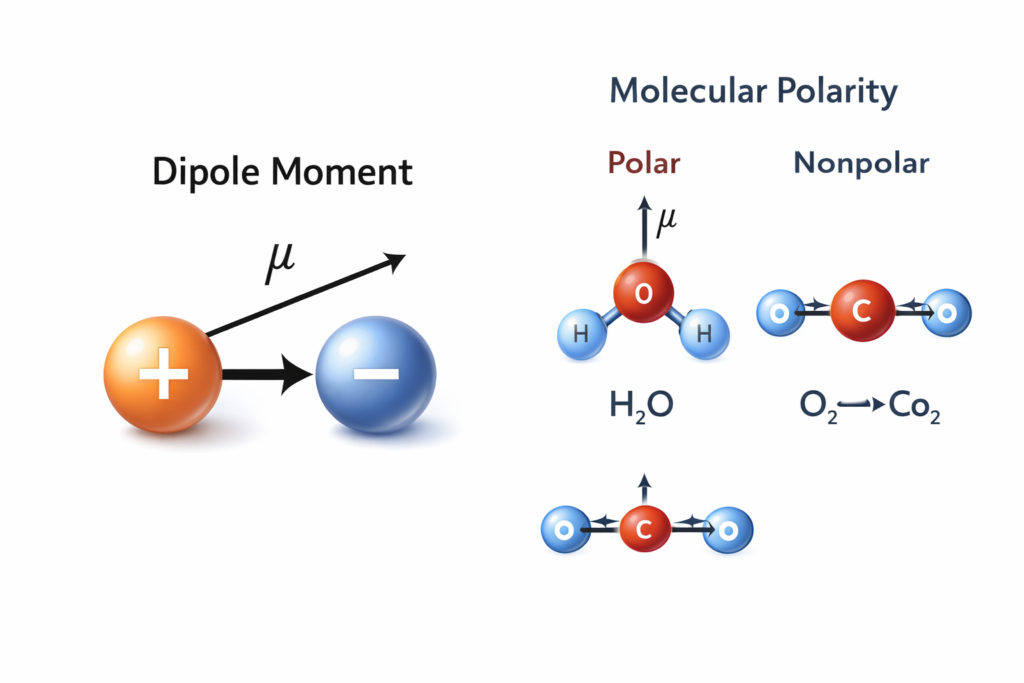
In simple terms, the dipole moment (μ) measures the separation of positive and negative electrical charges within a system. Whenever two opposite charges are separated by a certain distance, a dipole moment is created. The magnitude of this dipole moment depends on two main factors:
⚡ Magnitude of charge (q)
📏 Distance between charges (r)
The mathematical formula used to calculate dipole moment is: μ = q × r
Where:
μ = Dipole moment
q = magnitude of electric charge
r = distance between the charges
The SI unit of dipole moment is Coulomb-meter (C·m).
In chemistry, dipole moment is often used to determine whether a molecule is polar or non-polar. Polar molecules have unequal charge distribution, resulting in a non-zero dipole moment. Nonpolar molecules have symmetrical charge distribution, giving them a dipole moment close to zero.
For example:
🧪 Water (H₂O) has a strong dipole moment due to its bent molecular structure.
🧪 Carbon dioxide (CO₂) has zero dipole moment because its linear geometry cancels charge separation.
Understanding dipole moment is extremely important in many fields including:
🔬 Physical Chemistry
⚡ Electrostatics
🧬 Molecular Biology
💊 Pharmaceutical Research
🌍 Environmental Chemistry
Students studying Class 11 Chemistry, Class 12 Chemistry, and undergraduate physics or chemistry courses frequently encounter dipole moment calculations when learning about molecular geometry and bonding theory.
Traditionally, calculating dipole moment requires manual calculation and unit conversions, which can sometimes lead to errors. That is why the Dipole Moment Calculator is extremely useful. It automates the calculation and instantly displays the result in scientific notation using proper exponent format, making it easier for students to understand.
The calculator takes the charge magnitude and the separation distance as inputs and immediately computes the dipole moment using the formula μ = q × r.
This type of format is widely used in scientific research papers, textbooks, and laboratory experiments.
In molecular chemistry, dipole moment also helps determine electronegativity differences between atoms. When atoms with different electronegativity values bond together, electrons are not shared equally. This leads to partial charges on atoms and generates a dipole moment.
For example: ⚡ Hydrogen chloride (HCl) has a dipole moment because chlorine is more electronegative than hydrogen.
Dipole moment also plays an important role in determining intermolecular forces, including:
💧 Hydrogen bonding
⚡ Dipole-dipole interaction
🌫 Van der Waals forces
These forces affect many real-world properties such as:
Boiling point
Melting point
Solubility
Surface tension
In spectroscopy and advanced chemistry, dipole moment is also used in:
🔬 Infrared spectroscopy
📡 Microwave spectroscopy
🧬 Molecular structure analysis
Because of its wide range of applications, understanding dipole moment is essential for science students, researchers, engineers, and educators.
Using this free online Dipole Moment Calculator, students can instantly compute dipole moment values and verify their homework, assignments, or research calculations. The calculator simplifies complex concepts and makes learning physics and chemistry much more interactive and engaging.
If you are studying electrostatics, molecular polarity, chemical bonding, or physical chemistry, this tool will save time and improve calculation accuracy.
🌍Applications of Dipole Moment in Daily Life ⚙️
Although dipole moment is a microscopic property, its applications extend deeply into everyday life and advanced technology:
🔹 Microwave Ovens 🍽️: Polar molecules (like water) absorb microwave radiation due to their dipole moments, generating heat efficiently.
🔹 Pharmaceutical Drug Design 💊: Dipole moment influences drug–receptor interactions, solubility, and bioavailability.
🔹 Material Science & Polymers 🧱: Dielectric properties of materials depend on molecular dipole moments.
🔹 Atmospheric & Environmental Chemistry 🌫️: Pollutant behavior, greenhouse gas absorption, and climate modeling rely on dipole moment data.
🔹 Electronics & Semiconductors 💡: Dipole alignment affects dielectric constants, capacitors, sensors, and memory devices.
🔹 Spectroscopy & Analytical Chemistry 🔬: Infrared (IR) activity of molecules directly depends on dipole moment variation.
📌 Disclaimer ⚠️
📢 This Dipole Moment Calculator is designed for educational and informational purposes only. While the calculator provides accurate results based on standard scientific formulas, users should verify calculations when performing laboratory experiments, academic research, or professional scientific analysis.
This tool simplifies theoretical calculations but does not replace professional scientific software or laboratory instrumentation. Always consult textbooks, scientific references, or subject experts when conducting advanced chemical or physics research.
❓Frequently Asked Questions 🧠
Q1. What is a dipole moment?
A dipole moment is a vector quantity that measures the separation of positive and negative electrical charges within a molecule.
Q2. In which unit is dipole moment measured?🔎
Dipole moment is commonly measured in Debye (D) or Coulomb-meter (C·m).
Q3. Why is dipole moment important?
It helps determine molecular polarity, chemical reactivity, boiling point, and intermolecular interactions.
Q4. Can a molecule have zero dipole moment?
Yes, symmetrical molecules like CO₂ or BF₃ have zero net dipole moment despite polar bonds.
Q5. Is this calculator suitable for complex molecules?📈
It provides approximate values and is best suited for basic molecular systems and conceptual understanding.
Related Calculator
