⚛️ de Broglie Wavelength Calculator
h = 6.626 × 10-34 J·s
Table of Contents
✍️ Author & Academic Authority: Dr. Nitish Kr. Bharadwaj
📘 Qualifications: B.Sc., B.Ed., M.Sc., Ph.D. (Biochemistry), MBA (Financial Management)
🔬 de Broglie Wavelength Calculator – Introduction
Instantly Calculate Matter Wave Wavelength ⚛️
The de Broglie Wavelength Calculator is an advanced physics calculator designed to determine the wavelength associated with a moving particle using the famous de Broglie equation. This concept forms a fundamental pillar of quantum mechanics, explaining how particles such as electrons, protons, atoms, and even molecules exhibit wave-like properties. 🌊⚛️
In classical physics, matter and waves were considered completely different entities. However, in 1924, the French physicist Louis de Broglie revolutionized physics by proposing the idea that all matter possesses wave characteristics. This theory is now known as the de Broglie hypothesis, and the wavelength associated with a moving particle is called the de Broglie wavelength.
The mathematical expression for this phenomenon is: λ = h / p
Where:
- λ (lambda) = de Broglie wavelength
- h = Planck’s constant (6.626 × 10⁻³⁴ Js)
- p = momentum of the particle
Since momentum p = mv, the equation can also be written as: λ = h / mv
Where:
- m = mass of the particle
- v = velocity of the particle
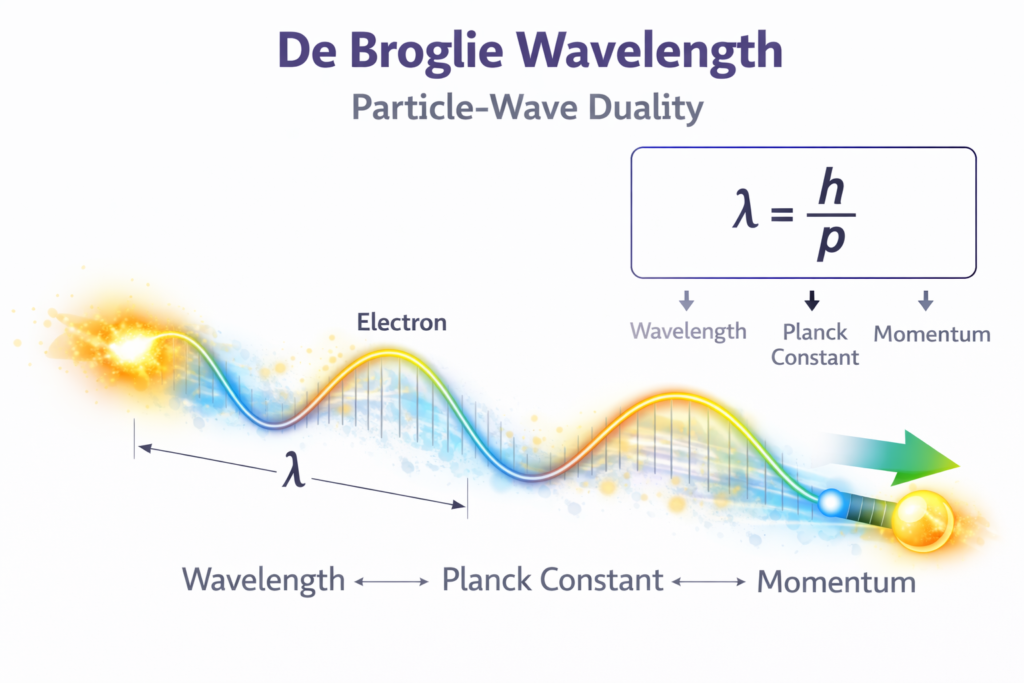
Our de Broglie wavelength calculator allows students, researchers, and educators to quickly compute the wavelength of matter waves by entering simple values such as mass and velocity or momentum. This removes the need for manual calculations and reduces the chances of errors during physics problem solving. 📊✨
🌌 Understanding Matter Waves in Quantum Physics
The wave-particle duality principle states that every particle exhibits both particle-like and wave-like behavior. The de Broglie wavelength formula explains this wave nature quantitatively.
For example:
- Fast-moving electrons behave like waves in electron microscopes. 🔬
- Neutrons and atoms can create interference patterns.
- Even larger molecules can demonstrate wave properties under controlled conditions.
However, the de Broglie wavelength of large objects such as cars or humans is extremely small because their mass is very large. Therefore, the wave behavior of macroscopic objects is practically impossible to observe.
This concept became one of the foundations of modern quantum mechanics, influencing theories such as:
- Schrödinger wave equation
- Heisenberg uncertainty principle
- Quantum tunneling
- Electron diffraction
⚙️ How the de Broglie Wavelength Calculator Works
The online de Broglie wavelength calculator automates the equation by applying the correct formula instantly.
Steps typically include:
1️⃣ Enter the mass of the particle
2️⃣ Enter the velocity or momentum
3️⃣ Click calculate
4️⃣ The calculator returns the de Broglie wavelength
Because the formula involves very small numbers, especially when using Planck’s constant, manual calculation often leads to rounding errors. This tool eliminates that issue and provides high-precision physics results instantly.
This makes the calculator particularly useful for:
🎓 Physics students
👨🏫 Teachers and educators
🧪 Researchers in quantum mechanics
📚 Competitive exam preparation
⚙️ How the de Broglie Wavelength Calculator Works
The online de Broglie wavelength calculator automates the equation by applying the correct formula instantly.
Steps typically include:
1️⃣ Enter the mass of the particle
2️⃣ Enter the velocity or momentum
3️⃣ Click calculate
4️⃣ The calculator returns the de Broglie wavelength
Because the formula involves very small numbers, especially when using Planck’s constant, manual calculation often leads to rounding errors. This tool eliminates that issue and provides high-precision physics results instantly.
This makes the calculator particularly useful for:
🎓 Physics students
👨🏫 Teachers and educators
🧪 Researchers in quantum mechanics
📚 Competitive exam preparation
🧪 Example Calculation
Suppose an electron moves at velocity 2 × 10⁶ m/s.
Using the equation: λ = h / mv
Where:
- h = 6.626 × 10⁻³⁴ Js
- m (electron mass) = 9.11 × 10⁻³¹ kg
- v = 2 × 10⁶ m/s
After calculation, the wavelength comes out to be approximately 3.64 × 10⁻¹⁰ meters, which lies in the X-ray wavelength region.
This explains why electron microscopes can produce extremely high-resolution images compared to optical microscopes.
🎓 Importance of the de Broglie Wavelength Concept
The de Broglie wavelength formula is one of the most important equations in quantum physics. It bridges the gap between classical mechanics and wave theory, proving that matter is not purely particulate.
Key reasons why this concept is important:
✔ Explains wave nature of electrons
✔ Supports quantum mechanical models of atoms
✔ Helps understand electron diffraction experiments
✔ Forms the basis of modern quantum technologies
Without the de Broglie hypothesis, many modern technologies such as electron microscopes, semiconductor devices, and nanotechnology would not exist.
🌍 Applications in Human Daily Life ⚛️
Although de Broglie wavelength calculations belong to quantum physics, they indirectly power many technologies used in everyday life.
🔬 Electron Microscopes: The de Broglie wavelength of electrons allows scientists to create electron microscopes, which can magnify objects up to 10 million times. These microscopes are essential for studying viruses, bacteria, and nanomaterials.
💻 Semiconductor Technology: Quantum principles including matter wave theory are fundamental to transistors and microchips, which power smartphones, laptops, and modern electronics.
🧬 Nanotechnology: The behavior of particles at extremely small scales depends on quantum wave properties. Engineers use these principles to design nanomaterials, quantum dots, and advanced sensors.
🧪 Particle Accelerators: Scientists calculate de Broglie wavelengths of high-speed particles to study fundamental particles in research facilities such as particle accelerators.
🛰 Advanced Imaging Technologies: Medical imaging technologies and scientific instruments rely on quantum wave behavior of particles to improve accuracy and resolution.
⚠️ Disclaimer
This de Broglie wavelength calculator is provided for educational and informational purposes only. While the calculator uses standard quantum mechanics formulas and physical constants, the results should be used as approximate references for learning and problem solving.
📚 Students should verify results during academic examinations or research calculations.
🔬 Scientific experiments may require high-precision instruments and controlled environments.
🧠 The calculator does not replace professional scientific analysis or laboratory measurements.
By using this tool, users agree that the website allcalculators.co.in is not responsible for decisions made based on calculated results.
📌 Related Calculator
❓FAQs
❓ What is the de Broglie wavelength?
The de Broglie wavelength is the wavelength associated with a moving particle according to quantum mechanics. It describes the wave nature of matter and is calculated using the formula λ = h/p.
❓ What is the formula for de Broglie wavelength?
The de Broglie wavelength formula is: λ = h / p or λ = h / mv; where h is Planck’s constant, m is mass, and v is velocity.
❓ Why is the de Broglie wavelength important?
The concept is essential for understanding wave-particle duality, electron diffraction, quantum mechanics, and technologies like electron microscopes and semiconductor devices.
❓ Can large objects have a de Broglie wavelength?
Yes. According to the de Broglie hypothesis, all objects have wavelengths. However, for large objects the wavelength is extremely small and impossible to observe.
❓ Who discovered the de Broglie wavelength?
The theory was proposed by Louis de Broglie in 1924, for which he later received the Nobel Prize in Physics in 1929.
