⚛️ Heisenberg’s Uncertainty Principle Calculator
Δx · Δp ≥ ħ / 2
Where ħ = h / 2π = 1.054 × 10-34 J·s
Table of Contents
✍️ Author & Academic Authority: Dr. Nitish Kr. Bharadwaj
📘 Qualifications: B.Sc., B.Ed., M.Sc., Ph.D. (Biochemistry), MBA (Financial Management)
🔬 Heisenberg’s Uncertainty Principle Calculator
Quantum physics is one of the most fascinating and mind-bending fields of science. At the heart of quantum mechanics lies a fundamental concept known as the Heisenberg Uncertainty Principle. This principle explains a deep limitation of nature: it is impossible to simultaneously know the exact position and exact momentum of a particle with perfect precision. Our Heisenberg Uncertainty Principle Calculator helps students, researchers, and physics enthusiasts instantly compute the uncertainty between position (Δx) and momentum (Δp) using the famous quantum mechanics equation.
In classical physics, objects behave predictably. If you know a car’s speed and position, you can calculate exactly where it will be later. However, at the quantum scale, particles such as electrons behave very differently. According to the uncertainty principle, the more precisely we measure the position of a particle, the less precisely we can measure its momentum, and vice versa.
The mathematical expression of the Heisenberg Uncertainty Principle is: Δx · Δp ≥ ħ / 2
Where:
Δx = Uncertainty in position
Δp = Uncertainty in momentum
ħ = Reduced Planck’s constant (h/2π)
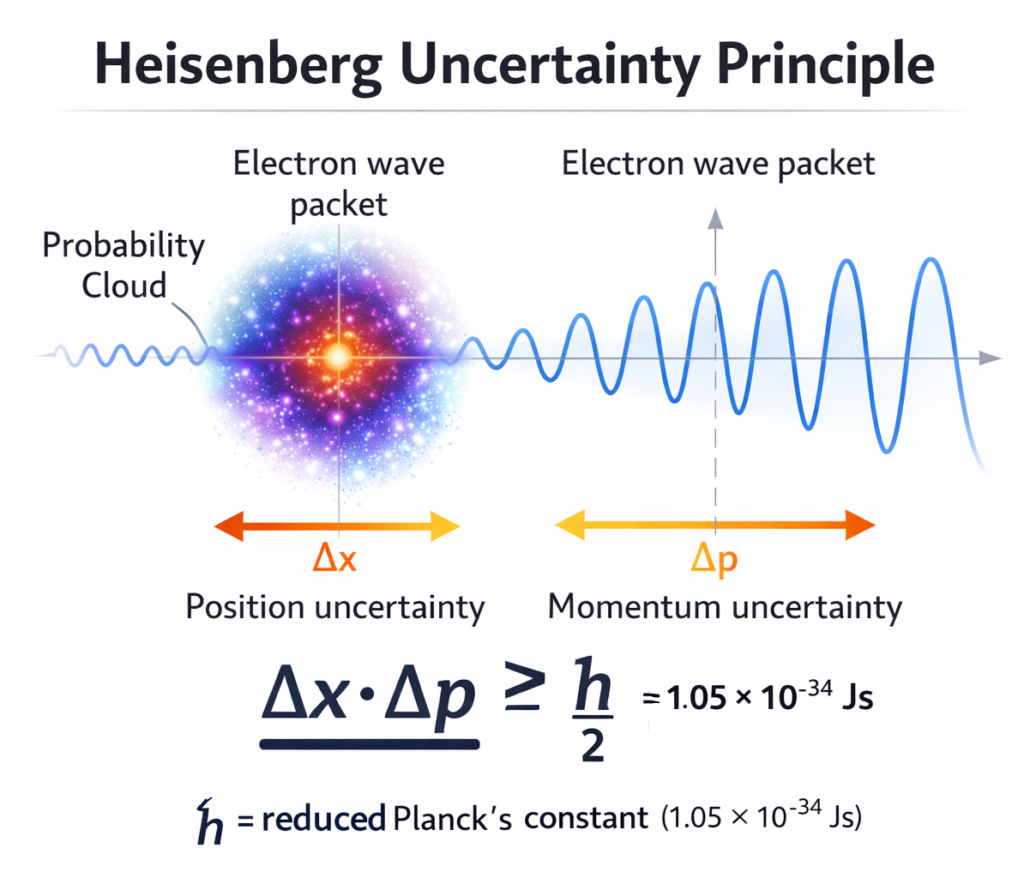
This equation shows that the product of uncertainties in position and momentum can never be smaller than a fundamental quantum limit. This limit arises not because of measurement errors, but because of the intrinsic nature of quantum particles.
Our online uncertainty principle calculator simplifies these calculations. Instead of manually rearranging formulas or performing complex computations, users can simply input the known values and instantly compute the unknown variable. This tool is especially helpful for:
🎓 Physics students solving homework or exam problems
👨🔬 Researchers studying quantum mechanics concepts
📚 Teachers explaining quantum physics interactively
🧠 Learners exploring the strange behavior of particles
Understanding the Heisenberg Uncertainty Principle is essential for grasping how atoms and subatomic particles behave. In the quantum world, electrons do not orbit the nucleus like planets around the sun. Instead, they exist in probability clouds. The uncertainty principle explains why electrons cannot collapse into the nucleus — their position cannot be perfectly defined without drastically increasing momentum uncertainty.
This principle was introduced in 1927 by German physicist Werner Heisenberg, one of the pioneers of quantum mechanics. His work revolutionized physics and fundamentally changed our understanding of reality. Today, the uncertainty principle plays a critical role in many advanced scientific fields including:
⚛️ Quantum mechanics
🔬 Nanotechnology
💻 Quantum computing
🧪 Atomic physics
🌌 Particle physics
Our Heisenberg Uncertainty Principle Calculator is designed with simplicity and accuracy in mind. It uses the precise value of Planck’s constant and applies the correct quantum mechanics formula to generate reliable results instantly.
With this calculator, users can determine:
📍 Minimum uncertainty in position (Δx)
🚀 Minimum uncertainty in momentum (Δp)
📐 Quantum mechanical limits of measurement
This is extremely useful when solving physics numerical problems, preparing for competitive exams like JEE, NEET, SAT Physics, and university-level quantum mechanics courses.
Another advantage of using an online quantum physics calculator is that it eliminates calculation mistakes. Quantum formulas often involve extremely small constants (like Planck’s constant ≈ 6.626 × 10⁻³⁴ Js). Manual calculations with such tiny numbers can easily lead to errors, but automated calculators handle them accurately.
Beyond academics, the Heisenberg Uncertainty Principle has deep philosophical implications. It shows that nature itself has fundamental limits on knowledge. Even with perfect instruments, we cannot measure everything exactly at the same time. This concept reshaped scientific thinking and influenced modern interpretations of reality.
Today, this principle is essential in designing technologies such as:
🔬 Electron microscopes
💾 Quantum computers
📡 Semiconductor devices
🧪 Particle accelerators
By using our Heisenberg Uncertainty Principle Calculator, you can instantly explore how changing the uncertainty in one variable affects the other. This interactive learning approach makes quantum physics easier to understand and visualize.
Whether you’re solving homework problems, preparing for exams, or simply curious about the strange world of quantum mechanics, this uncertainty principle calculator provides fast, accurate, and reliable results in seconds.
🌍 Applications in Daily Life ⚛️
Although the Heisenberg Uncertainty Principle originates in quantum mechanics, its implications influence many technologies used in everyday life.
🔬 Electron Microscopes: Electron microscopes rely on quantum physics to observe extremely small structures like viruses and atoms. The uncertainty principle determines the resolution limits of these instruments.
💻 Quantum Computing: Modern quantum computers use quantum mechanical principles including uncertainty to process information in new ways. These machines could revolutionize cryptography, AI, and scientific simulations.
📱 Semiconductor Electronics: Devices like smartphones, computers, and microchips rely on semiconductor physics. Quantum uncertainty affects how electrons behave inside transistors.
🧪 Nanotechnology: At the nanoscale, the position and momentum of particles become critical. Engineers must account for uncertainty effects when designing nano-devices and materials.
🛰️ Particle Physics Experiments: Large facilities such as particle accelerators study subatomic particles where the uncertainty principle calculator helps predict measurement limits.
⚠️ Disclaimer 📢
The Heisenberg Uncertainty Principle Calculator available on this website is designed for educational and informational purposes only. While we strive to ensure that the calculator uses accurate physics formulas and constants, the results should be used as a learning aid rather than a substitute for professional scientific calculations or academic verification.
📚 Students are encouraged to understand the underlying quantum mechanics formulas and principles instead of relying solely on automated tools.
🔬 Researchers and professionals should verify results using validated computational methods or scientific software.
By using this online uncertainty principle calculator, you acknowledge that the website owners are not responsible for academic, research, or engineering decisions made based on the calculated results.
📌 Related Calculator
❓FAQs
❓ What is the Heisenberg Uncertainty Principle?
The Heisenberg Uncertainty Principle is a fundamental rule of quantum mechanics stating that the exact position and momentum of a particle cannot both be known simultaneously with perfect accuracy.
❓ What is the formula of the uncertainty principle?
The formula is: Δx · Δp ≥ ħ / 2
Where Δx is uncertainty in position, Δp is uncertainty in momentum, and ħ is the reduced Planck constant.
❓ How does the Heisenberg Uncertainty Principle Calculator work?
The calculator uses the quantum mechanics uncertainty equation to determine the minimum uncertainty of either position or momentum when the other value is known.
❓ Why is the uncertainty principle important in physics?
The principle explains the behavior of particles at the quantum level and is essential for understanding atomic structure, quantum mechanics, and modern technologies like semiconductors and quantum computing.
❓ Does the uncertainty principle apply to large objects?
In theory yes, but for macroscopic objects the uncertainties are extremely small, so classical physics works perfectly for everyday objects.
