⚛️ Quantum Number Calculator
Principal Quantum Number:
n = 1,2,3,4…
Angular Momentum Quantum Number:
l = 0 → (n − 1)
Magnetic Quantum Number:
m = −l → +l
Spin Quantum Number:
s = +1/2 or −1/2
Table of Contents
✍️ Author & Academic Authority: Dr. Nitish Kr. Bharadwaj
📘 Qualifications: B.Sc., B.Ed., M.Sc., Ph.D. (Biochemistry), MBA (Financial Management)
🔬 Quantum Number Calculator – Introduction
Find Electron Quantum Numbers Instantly ⚛️
Understanding the quantum numbers of electrons is one of the most fundamental concepts in atomic physics and quantum mechanics. However, calculating the four quantum numbers manually for different orbitals and electrons can be confusing and time-consuming for students. This is exactly why our Quantum Number Calculator is designed — to help you instantly determine the principal, azimuthal, magnetic, and spin quantum numbers of an electron with complete accuracy.
This online quantum number calculator simplifies complex quantum mechanics calculations into an easy and interactive tool. Whether you are a high school student, college learner, physics enthusiast, or chemistry student, this calculator allows you to quickly determine the quantum state of electrons inside atoms.
Quantum numbers are a set of numerical values that describe the position and energy of an electron in an atom. According to quantum mechanics, electrons do not orbit the nucleus in simple circular paths like planets around the sun. Instead, they exist in probability regions called atomic orbitals. Quantum numbers help describe these orbitals and the properties of electrons within them.
There are four quantum numbers used to describe the state of an electron:
⚛️ Principal Quantum Number (n)
⚛️ Azimuthal Quantum Number (l)
⚛️ Magnetic Quantum Number (m)
⚛️ Spin Quantum Number (s)
Each quantum number provides unique information about the electron.
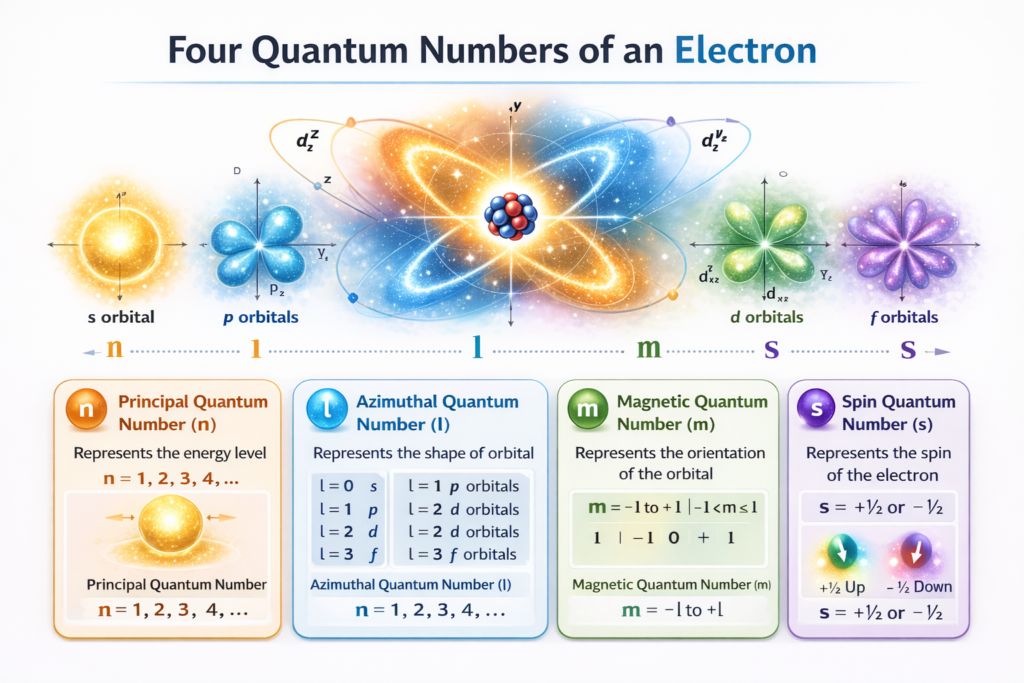
The principal quantum number (n) describes the main energy level or shell in which the electron resides. The value of n is always a positive integer such as 1, 2, 3, 4, 5…. As the value of n increases, the electron moves farther from the nucleus and the energy of the orbital increases.
The azimuthal quantum number (l) determines the shape of the orbital and the subshell where the electron exists. The value of l ranges from 0 to (n − 1). These values correspond to different subshells:
0 → s orbital
1 → p orbital
2 → d orbital
3 → f orbital
These orbitals determine the electron cloud distribution around the nucleus.
The magnetic quantum number (m) describes the orientation of an orbital in space. Its value ranges from −l to +l, including zero. This means that for each subshell, multiple orbitals can exist with different spatial orientations.
Finally, the spin quantum number (s) represents the direction of electron spin. Electrons behave like tiny magnets and can spin in two possible directions:
+1/2 → Spin up
−1/2 → Spin down
According to the Pauli Exclusion Principle, no two electrons in an atom can have the same set of all four quantum numbers.
Understanding these four numbers is essential for mastering electron configuration, atomic structure, and quantum mechanics.
However, determining them manually for different electrons often requires careful analysis of electron configurations and orbital diagrams. This is where an online Quantum Number Calculator becomes extremely useful.
Our calculator allows users to input electron configuration information or orbital details and instantly determine the corresponding n, l, m, and s values.
This tool is particularly useful for solving problems in:
📚 Atomic physics
🧪 Chemistry electron configuration
🎓 Quantum mechanics assignments
📖 Competitive exams (JEE, NEET, SAT, etc.)
Students frequently struggle with questions like:
“Find the four quantum numbers for the last electron in an atom.”
“Determine the quantum numbers for an electron in the 3p orbital.”
“Calculate n, l, m, s for a given electron configuration.”
Using the Quantum Number Calculator, you can obtain these values within seconds without making calculation mistakes.
The calculator is built with accurate quantum mechanics rules, ensuring reliable results every time.
Another major advantage of using this calculator is visual learning. When students interact with computational tools, they understand atomic structure concepts much faster compared to traditional memorization.
Quantum numbers are not only important in physics but also play a major role in modern technology and scientific research.
Fields like:
🔬 quantum computing
🧬 spectroscopy
⚡ semiconductor physics
🛰️ nanotechnology
all rely heavily on quantum mechanics principles.
Electrons in atoms determine the chemical properties of elements, and quantum numbers describe exactly how those electrons behave. This is why quantum numbers are the foundation of modern atomic theory.
The Quantum Number Calculator on AllCalculators.co.in is designed to help students and learners quickly explore these concepts in an interactive way.
Instead of spending time solving repetitive calculations, students can focus on understanding the physics behind the numbers.
Our calculator works instantly and requires no complex formulas to remember. Simply input the required information, and the tool calculates the correct electron quantum numbers automatically.
This tool is also mobile-friendly, meaning students can use it during study sessions, homework solving, or exam preparation directly from their smartphones.
Because the tool is optimized for speed and simplicity, it can significantly improve productivity when solving physics or chemistry problems. The calculator is also extremely helpful for teachers who want to demonstrate quantum mechanics concepts in classrooms using interactive digital tools.
At AllCalculators.co.in, our goal is to make complex educational concepts simple and accessible to everyone. The Quantum Number Calculator is part of our growing collection of physics calculators designed to help students learn faster and solve problems efficiently.
Whether you are learning atomic structure for the first time or preparing for advanced exams, this tool will help you calculate electron quantum numbers quickly, accurately, and effortlessly. Start using the Quantum Number Calculator today and explore the fascinating world of quantum mechanics and atomic physics with ease.
🌍 Applications in Daily Life
Although quantum numbers seem purely theoretical, they play an essential role in many technologies used in everyday life.
⚡ Electronics & Semiconductors: Quantum numbers determine electron energy levels in atoms, which directly affects how materials conduct electricity. Modern semiconductors, computer chips, and transistors rely on quantum mechanics principles.
💡 LED Lights and Lasers: Quantum transitions between electron energy levels produce light. Devices like LED bulbs, laser pointers, and fiber optic communication systems work because of these quantum effects.
📱 Smartphones and Microprocessors: The design of microprocessors and integrated circuits depends on understanding electron behavior in atoms and materials.
🧪 Chemical Reactions: Quantum numbers explain how atoms bond together, which is fundamental to chemistry, pharmaceuticals, and material science.
🧬 Medical Imaging: Technologies such as MRI scanners use principles related to quantum spin states of particles.
🛰️ Quantum Computing: Future computing systems rely on quantum bits (qubits) based on electron spin and quantum states.
Thus, the concept behind this Quantum Number Calculator is deeply connected to modern scientific and technological developments.
⚠️ Disclaimer 📢
The Quantum Number Calculator available on AllCalculators.co.in is designed for educational and informational purposes only.
While this calculator uses accurate quantum mechanics formulas and atomic physics principles, the results should be used primarily as a learning aid for students, teachers, and academic problem solving.
Users are encouraged to verify results when using the tool for academic assignments, research work, or examination preparation.
Although we strive to maintain high accuracy and reliability, AllCalculators.co.in does not guarantee that every calculation result will be completely error-free under all conditions.
By using this online physics calculator, you acknowledge that the results are intended for study support and conceptual understanding.
📌 Related Calculator
❓FAQs
❓ What are the four quantum numbers of an electron?
The four quantum numbers are principal quantum number (n), azimuthal quantum number (l), magnetic quantum number (m), and spin quantum number (s). These values describe the energy, shape, orientation, and spin of an electron in an atom.
❓ What does the principal quantum number represent?
The principal quantum number (n) represents the main energy level or shell of an electron around the atomic nucleus.
❓ Can two electrons have the same quantum numbers?
According to the Pauli Exclusion Principle, two electrons cannot have the same set of all four quantum numbers within an atom.
❓ How does a Quantum Number Calculator work?
A Quantum Number Calculator determines the values of n, l, m, and s using rules of quantum mechanics and electron configuration.
❓ Why are quantum numbers important in physics and chemistry?
Quantum numbers explain electron structure, atomic behavior, chemical bonding, and energy levels, which are fundamental concepts in physics and chemistry.
