🔥 Carnot Efficiency Calculator
Table of Contents
✍️ Author & Academic Authority: Dr. Nitish Kr. Bharadwaj
📘 Qualifications: B.Sc., B.Ed., M.Sc., Ph.D. (Biochemistry), MBA (Financial Management)
🔥 Carnot Efficiency Calculator – Introduction
Instantly Calculate Maximum Heat Engine Efficiency 📊
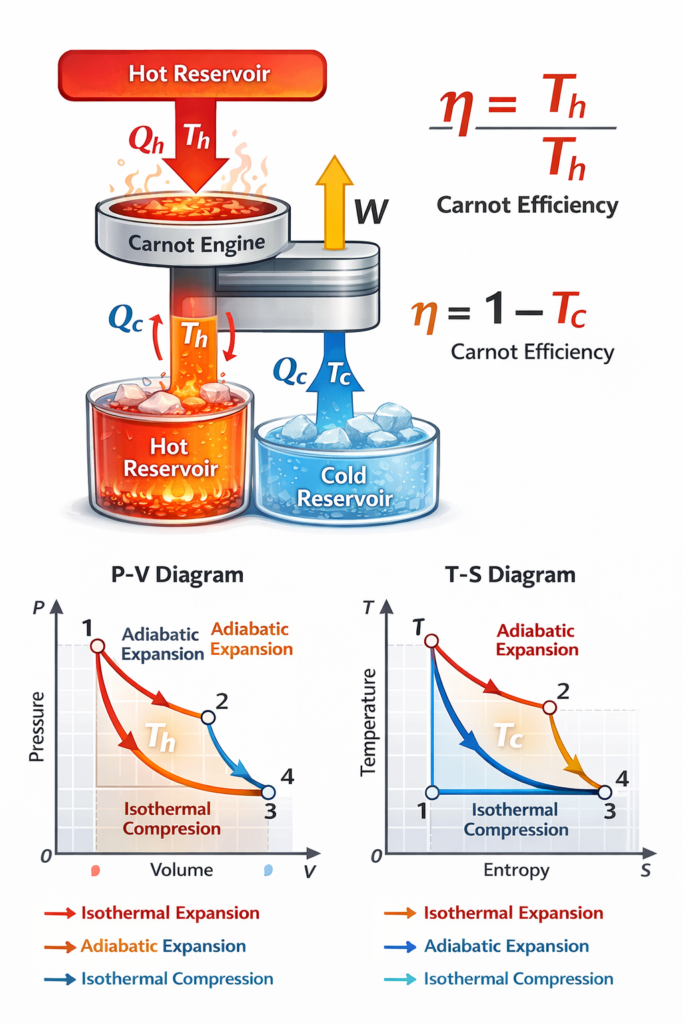
The Carnot Efficiency Calculator is a powerful physics tool designed to help students, engineers, and science enthusiasts calculate the maximum theoretical efficiency of a heat engine operating between two temperatures. This concept comes from thermodynamics, specifically the Carnot cycle, which represents the most efficient possible engine operating between a hot and cold reservoir.
In simple terms, the Carnot efficiency formula determines the highest possible efficiency any heat engine can achieve when converting heat energy into useful work. No real engine can exceed this efficiency because it would violate the second law of thermodynamics. That’s why the Carnot efficiency equation is considered a fundamental limit for engine performance.
The Carnot efficiency formula is: η = 1 − (Tc / Th)
Where:
η = Carnot efficiency
Tc = Temperature of the cold reservoir (in Kelvin)
Th = Temperature of the hot reservoir (in Kelvin)
This formula tells us that the efficiency of a heat engine increases when:
• The temperature of the hot reservoir increases
• The temperature of the cold reservoir decreases
However, both temperatures must always be expressed in Kelvin, not Celsius or Fahrenheit, for accurate thermodynamic calculations.
Our Carnot Efficiency Calculator online simplifies this calculation instantly. Instead of manually solving the formula, users only need to enter the hot reservoir temperature (Th) and the cold reservoir temperature (Tc). The calculator immediately determines the maximum theoretical efficiency percentage.
This online physics calculator is especially helpful for students studying thermodynamics, physics, mechanical engineering, and energy systems. Many competitive exams and engineering problems involve heat engine efficiency calculations, making this tool extremely useful for quick verification of answers.
The concept of Carnot efficiency originates from Sadi Carnot, a French physicist who studied the limits of heat engines in the early 19th century. His theoretical model, known as the Carnot cycle, consists of four reversible processes:
- Isothermal expansion
- Adiabatic expansion
- Isothermal compression
- Adiabatic compression
These processes form an ideal cycle where no energy is lost due to friction, turbulence, or heat leakage. While real engines cannot achieve perfect Carnot efficiency, this theoretical model provides a benchmark for evaluating real-world engines such as:
• Steam engines
• Gas turbines
• Internal combustion engines
• Power plant turbines
Using a Carnot cycle efficiency calculator, students can quickly analyze how temperature differences influence engine performance. The larger the difference between the hot and cold reservoirs, the higher the possible efficiency.
For example:
Hot reservoir temperature (Th) = 600 K
Cold reservoir temperature (Tc) = 300 K
Carnot efficiency:
η = 1 − (300 / 600)
η = 0.5 or 50%
This means the maximum theoretical efficiency is 50%, and no engine operating between these temperatures can exceed that limit.
Understanding this concept is critical in engineering design, energy optimization, and thermal system analysis. Power plants, refrigeration systems, and turbines are all evaluated based on thermodynamic efficiency limits derived from Carnot’s theory.
Our free Carnot Efficiency Calculator removes the complexity of manual thermodynamics calculations and helps learners focus on understanding the physics principles behind heat engines.
Whether you are solving physics homework, preparing for competitive exams, or designing energy systems, this calculator provides accurate, instant, and reliable results.
Key advantages of using this calculator include:
⚡ Instant efficiency calculation
📚 Ideal for physics students and engineering learners
📊 Accurate thermodynamic formula implementation
🌍 Accessible anytime online
🧠 Helps understand Carnot cycle concepts
Because energy efficiency and thermal systems are increasingly important in modern technology, learning how to calculate Carnot efficiency helps engineers design more efficient engines, turbines, and power systems.
By using this Carnot efficiency calculator online, users can quickly determine the theoretical efficiency limit and better understand the thermodynamic principles governing heat engines.
🌍 Applications in Daily Life
Although the Carnot cycle is theoretical, the Carnot efficiency concept plays a major role in many real-world technologies.
🔥 Power Plants: Thermal power plants use heat to generate electricity. Engineers use Carnot efficiency calculations to determine the maximum efficiency limit of turbines and steam engines.
🚗 Automobile Engines: Car engines convert fuel heat into mechanical work. The heat engine efficiency formula helps engineers design engines with improved fuel efficiency.
❄️ Refrigerators & Air Conditioners: Cooling systems operate using reverse heat engine cycles. Understanding thermodynamic efficiency helps improve refrigeration performance and energy savings.
🏭 Industrial Energy Systems: Factories use boilers and turbines to produce energy. Engineers compare real engine efficiency vs Carnot efficiency to evaluate system performance.
🌞 Renewable Energy Systems: Solar thermal power plants rely on heat to generate electricity. Carnot efficiency calculations help determine the theoretical performance limits of these systems.
📚 Physics & Engineering Education: Students studying thermodynamics, mechanical engineering, and physics frequently use Carnot efficiency calculators to solve textbook problems and assignments.
⚠️ Disclaimer 📢
The Carnot Efficiency Calculator available on this website is designed for educational and informational purposes only. While the calculator uses the standard Carnot efficiency formula from thermodynamics, results are theoretical values representing the maximum possible efficiency of an ideal heat engine.
Real-world engines always operate below Carnot efficiency due to:
• Friction losses
• Heat leakage
• Irreversibility in processes
• Mechanical limitations
Users should not use this calculator as the sole basis for engineering design, industrial system planning, or professional thermodynamic analysis. Always consult qualified engineers or scientific resources when designing real heat engine systems.
By using this calculator, users agree that the website allcalculators.co.in is not responsible for any decisions or outcomes resulting from the use of calculated results.
📌 Related Calculator
❓ FAQs
❓ What is Carnot Efficiency in thermodynamics?
Carnot efficiency is the maximum theoretical efficiency a heat engine can achieve when operating between two temperature reservoirs. It is determined using the Carnot efficiency formula: η = 1 − (Tc / Th).
❓ How does a Carnot Efficiency Calculator work?
A Carnot Efficiency Calculator computes the theoretical efficiency of an ideal heat engine by using the temperatures of the hot reservoir and cold reservoir expressed in Kelvin.
❓ Why is Carnot efficiency the maximum possible efficiency?
According to the second law of thermodynamics, no engine operating between two heat reservoirs can be more efficient than a Carnot engine, because some energy must always be lost as waste heat.
❓ Why must temperatures be in Kelvin for Carnot efficiency calculations?
Thermodynamic equations require absolute temperature (Kelvin) because it starts from absolute zero, ensuring accurate energy and entropy calculations.
❓ Can real engines reach Carnot efficiency?
No. Real engines always operate below Carnot efficiency due to friction, heat losses, and irreversible processes.
