🧪 Gibbs Free Energy Calculator
Table of Contents
✍️ Author & Academic Authority: Dr. Nitish Kr. Bharadwaj
📘 Qualifications: B.Sc., B.Ed., M.Sc., Ph.D. (Biochemistry), MBA (Financial Management)
🧬 Biochemical Reaction Gibbs Free Energy Calculator — Introduction
⚡ Unlock the Energy Secrets of Life at the Cellular Level
Every single moment your body is alive, millions of biochemical reactions are firing inside your cells — breaking down glucose, synthesizing proteins, pumping ions, contracting muscles, and repairing DNA. But have you ever wondered: what decides whether a reaction in your body actually happens or not? 🤔 The answer lies in one of the most powerful and elegant concepts in all of science — Gibbs Free Energy (ΔG). Our Biochemical Reaction Gibbs Free Energy Calculator is your ultimate free online tool to instantly compute ΔG for any biochemical reaction, understand reaction spontaneity, and master the thermodynamic principles that govern all of life’s chemistry. 🌟
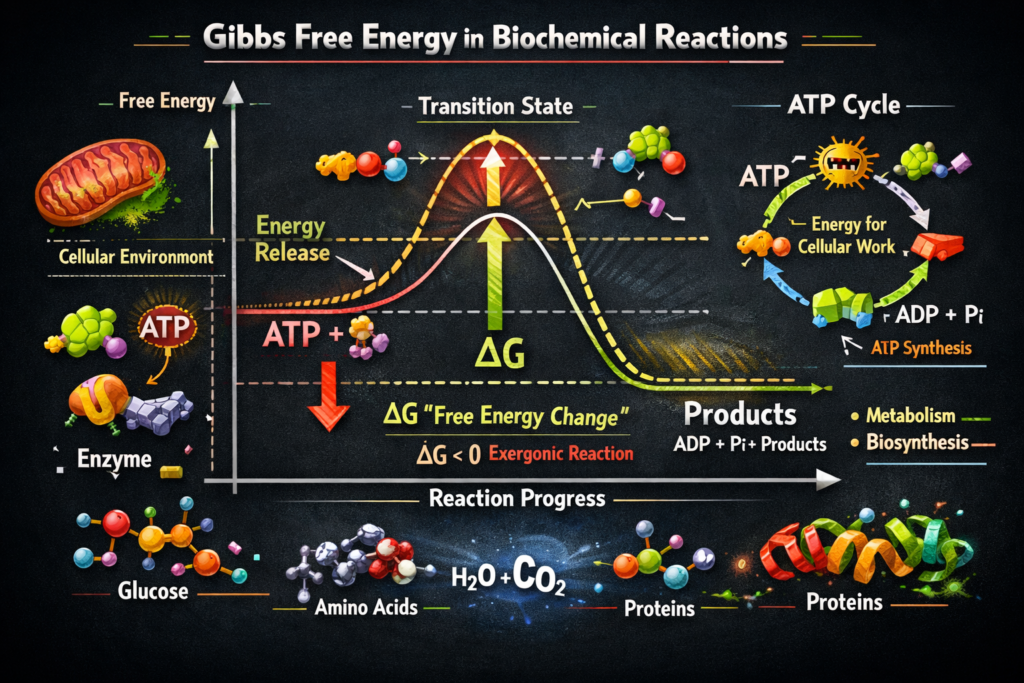
🔬 What is Gibbs Free Energy in Biochemistry?
Gibbs Free Energy — denoted by the symbol ΔG — is a thermodynamic state function that tells us the maximum amount of useful work a biochemical system can perform at constant temperature and pressure. Named after the legendary American physicist and mathematician Josiah Willard Gibbs (1839–1903), who first formulated this concept in the 1870s, the Gibbs free energy equation beautifully combines two fundamental properties of any chemical system: enthalpy (ΔH) and entropy (ΔS).
The master equation governing biochemical thermodynamics is: ΔG = ΔH − T·ΔS
Where:
🌡️ T = Absolute Temperature in Kelvin (K)
🌡️ ΔG = Change in Gibbs Free Energy (kJ/mol or J/mol)
🔥 ΔH = Change in Enthalpy (heat content of the system, kJ/mol)
🌀 ΔS = Change in Entropy (measure of molecular disorder, J/mol·K)
In biochemistry, a critically important modified form is the standard biochemical Gibbs free energy change (ΔG°’), measured at standard biological conditions: temperature of 298 K (25°C), pressure of 1 atm, pH 7.0, and all reactants and products at 1 M concentration. This distinction from the purely chemical standard state (pH 0) makes ΔG°’ the standard reference value used in virtually all biochemistry textbooks and laboratory research worldwide. 📚
⚡ Spontaneity: The Heart of Biochemical Reactions
The most critical application of Gibbs Free Energy in cell biology is the prediction of reaction spontaneity — whether a reaction will proceed forward on its own without needing external energy input. Here’s the universal rule that every biology and chemistry student must memorize:
- 🟢 ΔG < 0 (Negative) → The reaction is SPONTANEOUS (Exergonic) — it releases free energy and proceeds forward on its own. Example: Cellular respiration, ATP hydrolysis, glucose oxidation.
- 🔴 ΔG > 0 (Positive) → The reaction is NON-SPONTANEOUS (Endergonic) — it requires an input of energy to proceed. Example: Protein synthesis, gluconeogenesis, active transport.
- ⚖️ ΔG = 0 → The system is at EQUILIBRIUM — the forward and reverse reactions occur at equal rates, and there is no net change in reactant or product concentrations.
This is not merely an academic concept. Every living cell is governed by these thermodynamic rules, and no enzyme — however powerful — can make an energetically unfavorable reaction proceed. Enzymes accelerate reactions toward equilibrium, but they cannot change the direction determined by ΔG. This is a foundational principle of bioenergetics that bridges biochemistry, cell biology, and physiology. 🧫
🔗 ATP: The Cell’s Universal Energy Currency & Gibbs Free Energy
Perhaps the most celebrated biochemical application of Gibbs Free Energy is the hydrolysis of ATP (Adenosine Triphosphate) to ADP (Adenosine Diphosphate) and inorganic phosphate (Pᵢ): ATP + H₂O → ADP + Pᵢ ΔG°’ ≈ −30.5 kJ/mol
This highly negative ΔG°’ makes ATP hydrolysis a powerfully exergonic reaction, which is why ATP is the universal energy currency of all living cells. 💰 Cells couple energetically unfavorable (endergonic) reactions with ATP hydrolysis, making the overall coupled reaction spontaneous. This elegant biochemical strategy of coupled reactions is how cells build proteins, move muscles, synthesize DNA, pump molecules against concentration gradients, and perform every energy-requiring task essential for life.
The Krebs Cycle (Citric Acid Cycle), glycolysis, oxidative phosphorylation, and photosynthesis are all beautifully orchestrated networks of exergonic and endergonic reactions, each carefully regulated by the cell to maintain a negative net ΔG that keeps metabolic flux flowing in the right direction. 🔄
📊 The Reaction Quotient & Non-Standard Gibbs Free Energy
In real cellular conditions, reactant and product concentrations are never at the standard 1 M level. Cells operate under non-standard conditions, and the actual free energy change, ΔG, is related to the standard value by: ΔG = ΔG°’ + RT ln Q
Where R is the universal gas constant (8.314 J/mol·K), T is temperature in Kelvin, and Q is the reaction quotient — the ratio of actual product concentrations to reactant concentrations at the moment of measurement. This relationship means that cells can actually drive reactions in directions that seem thermodynamically unfavorable under standard conditions simply by manipulating concentrations of reactants and products — a profound insight that explains how metabolic pathways are dynamically regulated in living organisms. 🌐
🧪 Relationship Between ΔG and the Equilibrium Constant (Keq)
Another critical dimension of Gibbs Free Energy in biochemistry is its mathematical relationship with the equilibrium constant:
ΔG°’ = −RT ln Keq
This equation tells us that a large positive Keq (products heavily favored at equilibrium) corresponds to a large negative ΔG°’ (strongly spontaneous reaction), while a small Keq (reactants heavily favored) corresponds to a large positive ΔG°’ (strongly non-spontaneous). This relationship is used extensively in enzyme kinetics, pharmacology, metabolic engineering, and drug design. 💊
🏆 Why Use Our Biochemical Reaction Gibbs Free Energy Calculator?
Our free online ΔG Calculator at allcalculators.co.in is designed for students, researchers, educators, and science enthusiasts who need fast, accurate, and reliable Gibbs Free Energy calculations. Whether you’re solving biochemistry homework problems, preparing for NEET, JEE, MCAT, USMLE, or GRE Biology exams, conducting metabolic engineering research, or simply curious about the thermodynamics of life — this calculator is your perfect companion. 🎓
The calculator supports calculations using the standard ΔG = ΔH − TΔS formula and can handle inputs in multiple units (kJ/mol, J/mol, Kelvin, Celsius), automatically interpreting the sign and magnitude of ΔG to deliver a clear, plain-English spontaneity verdict. No complex manual math, no unit-conversion headaches — just instant, accurate results that help you understand the energy landscape of life’s chemistry. ⚡🔬
🌍 Applications in Human Daily Life
🔬 Understanding Metabolism & Energy Flow
This Gibbs free energy calculator helps students analyze how the body converts food into energy (ATP ⚡), making it essential for understanding metabolism and nutrition.
💊 Drug Development & Pharmaceuticals: Biochemists use ΔG calculations to predict whether drug interactions and biochemical reactions will occur efficiently inside the body.
🧠 Medical & Health Studies: Helps in understanding disease mechanisms where energy imbalance occurs, such as metabolic disorders.
🥗 Nutrition & Diet Planning: Understanding biochemical energy reactions helps explain how calories are utilized in the body.
🧪 Laboratory Research & Experiments: Researchers use biochemical reaction calculators to predict reaction feasibility before performing costly experiments.
🎓 Competitive Exam Preparation (NEET, JEE, CSIR-NET): Students can quickly solve thermodynamics-based biology and chemistry problems using this ΔG calculator.
🌱 Biotechnology & Bioengineering: Used in designing energy-efficient biochemical pathways in industrial and environmental applications.
⚠️ Disclaimer
⚠️ This Gibbs Free Energy Calculator for Biochemical Reactions is designed for educational and informational purposes only. While we strive to ensure high accuracy using standard thermodynamic equations, results may vary depending on input precision and real-world biological conditions 🧬.
📌 Users are advised to cross-check results for critical academic, medical, or research applications. This tool does not replace professional scientific analysis or laboratory validation.
🚀 By using this calculator, you agree that allcalculators.co.in is not liable for any decisions made based on calculated results.
📌 Related Calculator
❓ FAQs
❓ What is Gibbs Free Energy (ΔG) in biochemistry? ⚡
Gibbs free energy (ΔG) is a thermodynamic quantity that indicates whether a biochemical reaction is spontaneous or not. A negative ΔG means the reaction releases energy and occurs spontaneously.
❓ How do you calculate Gibbs free energy for a reaction? 🧪
You can calculate it using the formula: ΔG = ΔG° + RT ln Q. Our Gibbs free energy calculator simplifies this process instantly.
❓ What does a negative ΔG value mean? 📉
A negative ΔG indicates an exergonic reaction, meaning the reaction releases energy and can occur spontaneously.
❓ Why is Gibbs free energy important in cell biology? 🧬
It helps determine whether cellular processes like ATP synthesis and metabolic reactions can occur under physiological conditions.
❓ Can this calculator be used for exam preparation? 🎓
Yes! This ΔG calculator for biology and chemistry is ideal for NEET, JEE, and university-level exams.
