🧪 Osmolarity Calculator
Table of Contents
✍️ Author & Academic Authority: Dr. Nitish Kr. Bharadwaj
📘 Qualifications: B.Sc., B.Ed., M.Sc., Ph.D. (Biochemistry), MBA (Financial Management)
🧬 Osmolarity Calculator – Introduction
Calculate Solution Concentration Easily ⚗️
Osmolarity is a fundamental concept in cell biology, physiology, and biochemistry that determines how solutes influence water movement across cell membranes. Whether you’re a student solving textbook problems, a medical aspirant preparing for exams, or a lab professional working with solutions, understanding osmolarity is critical. That’s exactly where an Osmolarity Calculator becomes an essential tool 🔥.
👉 Osmolarity refers to the total concentration of all osmotically active particles in a solution, expressed as osmoles per liter (Osm/L). It plays a key role in maintaining cellular homeostasis, regulating fluid balance, and ensuring proper functioning of biological systems.
⚗️ What is Osmolarity?
Osmolarity is calculated using the formula: Osmolarity = Molarity × Number of particles (i)
Where:
- Molarity (M) = concentration of solute (mol/L)
- i (Van’t Hoff factor) = number of particles the compound dissociates into
For example:
- NaCl → dissociates into Na⁺ and Cl⁻ → i = 2
- Glucose → does not dissociate → i = 1
💡 This means electrolytes contribute more to osmolarity than non-electrolytes.
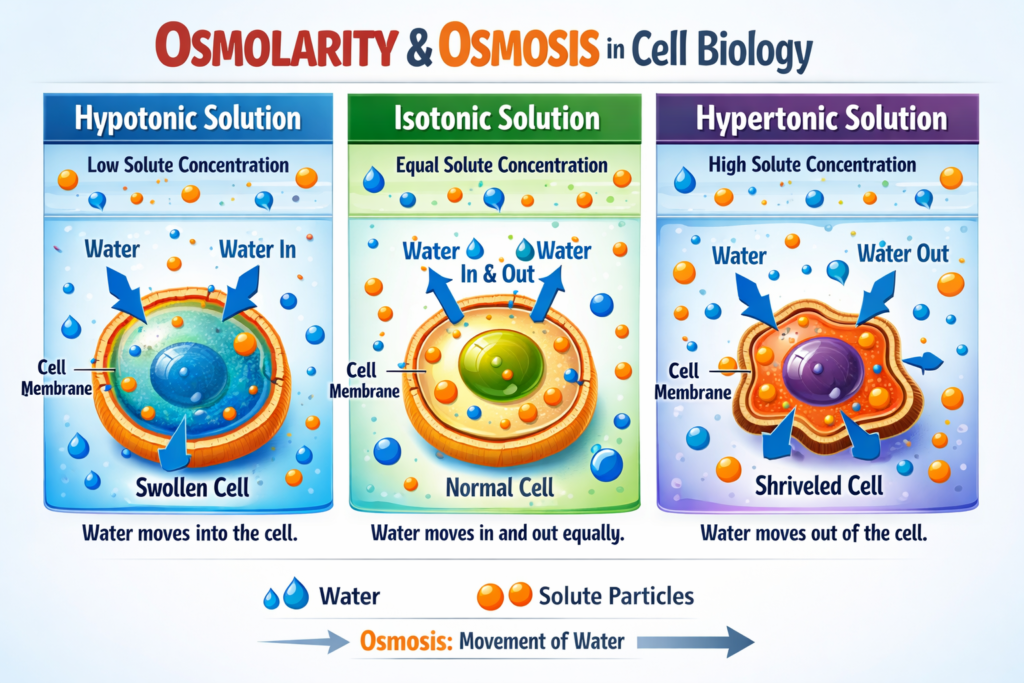
🧬 Why Osmolarity Matters in Biology?
In living organisms, osmolarity determines the direction of osmosis, the process where water moves across semipermeable membranes.
- If extracellular fluid is hypertonic, cells shrink 😨
- If it is hypotonic, cells swell and may burst 💥
- If it is isotonic, cells remain stable ✅
This balance is crucial in:
- Blood plasma regulation
- Kidney function
- IV fluid administration
- Cellular metabolism
⚡ Osmolarity vs Osmolality (Important Concept)
Many users confuse these two terms:
| Property | Osmolarity | Osmolality |
| Unit | Osmoles per liter (Osm/L) | Osmoles per kg (Osm/kg) |
| Depends on | Volume | Mass |
| Usage | Lab solutions | Clinical measurements |
👉 For most biological and lab purposes, osmolarity calculation is sufficient.
📊 Example Calculation
If you have a 1 M NaCl solution:
Osmolarity = 1 × 2 = 2 Osm/L
If you mix:
- 1 M glucose (i=1)
- 1 M NaCl (i=2)
Total Osmolarity = 1×1 + 1×2 = 3 Osm/L
🌍 Applications of Osmolarity in Daily Life 💧
Osmolarity is not just a textbook concept—it directly affects real-world biological and medical scenarios.
🏥 Medical Applications
- IV fluids are designed based on osmolarity balance to prevent cell damage 💉
- Used in diagnosing dehydration and electrolyte imbalance
- Critical in kidney and dialysis treatments
🍹 Food & Beverage Industry
- Used to control preservation and shelf-life
- Sugar concentration affects osmotic pressure in juices
🧴 Pharmaceuticals
- Drug formulations rely on correct osmolarity levels to ensure safe absorption
- Eye drops and injections must be isotonic 👁️
🌱 Agriculture
- Soil osmolarity impacts plant water absorption
- Helps in irrigation planning and fertilizer use
🧠 Academic & Competitive Exams
- Frequently asked in NEET, UPSC, and biology exams
- Essential for solving numerical problems quickly
⚠️ Disclaimer 🚨
📢 This Osmolarity Calculator is designed for educational and informational purposes only. While we strive for high accuracy, results may vary depending on input assumptions and real-world conditions.
❗ Always consult:
- Qualified healthcare professionals 🏥
- Laboratory experts 🧪
- Academic references 📚
before making critical medical or scientific decisions.
👉 Use this tool as a learning aid, not a substitute for professional judgment.
📌 Related Calculator
❓ FAQs – Osmolarity Calculator 🔥
❓ What is osmolarity in simple terms?
Osmolarity is the total concentration of dissolved particles in a solution that affect water movement across membranes.
❓ How do you calculate osmolarity quickly?
Use the formula: Osmolarity = Molarity × number of particles (i) or use an online osmolarity calculator for instant results.
❓ What is the difference between osmolarity and osmolality?
Osmolarity is based on volume (L), while osmolality is based on mass (kg).
❓ Why is osmolarity important in human body?
It helps maintain fluid balance, prevents cell damage, and supports vital functions like kidney regulation.
❓ Can osmolarity be negative?
No, osmolarity is always a positive value because it represents concentration.
