⚡ RMS Velocity Calculator
vrms = √(3RT / M)
Table of Contents
✍️ Author & Academic Authority: Dr. Nitish Kr. Bharadwaj
📘 Qualifications: B.Sc., B.Ed., M.Sc., Ph.D. (Biochemistry), MBA (Financial Management)
🚀 RMS Velocity Calculator – Introduction
Instantly Calculate Root Mean Square Velocity of Gas Molecules ⚛️
Understanding the motion of gas molecules is one of the most fascinating concepts in thermodynamics and kinetic theory of gases. The RMS Velocity Calculator is a powerful physics tool that helps students, researchers, and educators instantly calculate the Root Mean Square (RMS) velocity of gas molecules using the standard physics formula derived from kinetic molecular theory. RMS velocity represents the square root of the average of the squares of molecular speeds in a gas sample. In simpler terms, it provides an effective measure of the average molecular motion within a gas system.
In physics, especially in the kinetic theory of gases, gas molecules move randomly in all directions with different speeds due to continuous collisions with other molecules and container walls. Because the molecular speeds vary constantly, scientists use RMS velocity to represent the statistically meaningful average velocity of molecules. This value is extremely important in understanding how gases behave under different conditions of temperature, molar mass, and pressure.
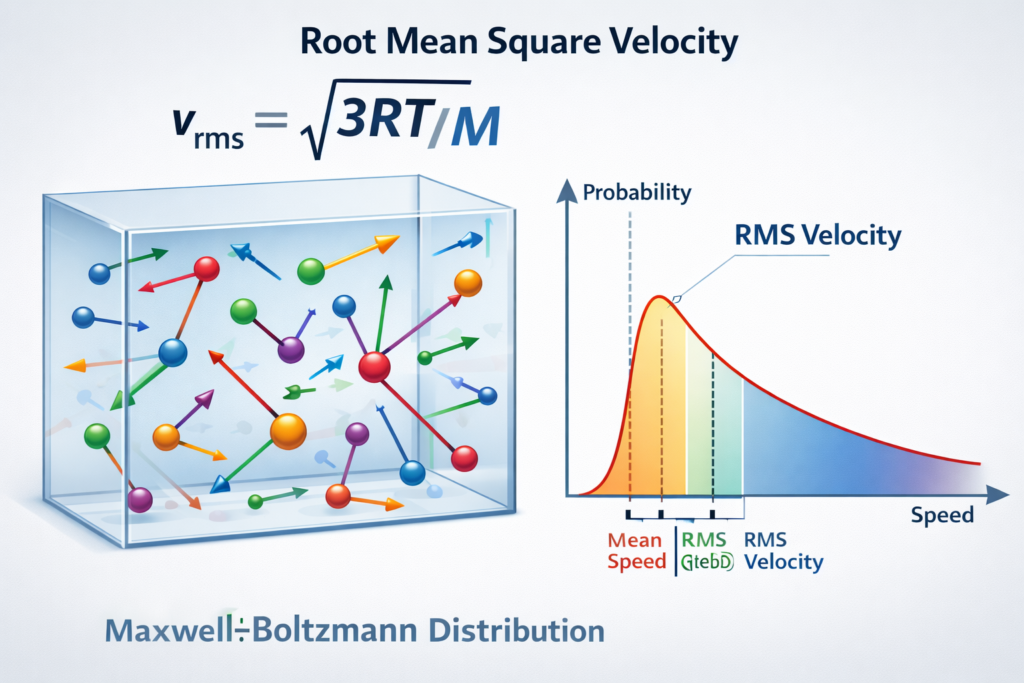
The RMS velocity formula is derived from kinetic energy relations and is expressed as: vᵣₘₛ = √(3RT / M)
Where:
• vᵣₘₛ = Root Mean Square Velocity
• R = Universal Gas Constant
• T = Absolute Temperature (Kelvin)
• M = Molar Mass of Gas
This formula clearly shows that temperature plays a crucial role in determining molecular speed. When temperature increases, molecules gain kinetic energy and move faster, which increases the RMS velocity. On the other hand, gases with larger molar mass move more slowly because heavier molecules require more energy to accelerate.
The RMS Velocity Calculator simplifies this entire process. Instead of performing manual calculations, users can simply input the temperature of the gas and its molar mass, and the calculator will instantly determine the root mean square speed of gas molecules. This tool is particularly helpful for students preparing for physics exams, engineering aspirants, and researchers working in thermodynamics or molecular physics.
One of the biggest advantages of using an online RMS speed calculator is the elimination of calculation errors. When solving physics problems manually, students often make mistakes in square roots, unit conversions, or formula substitution. An automated calculator ensures accurate and instant results, allowing learners to focus more on conceptual understanding rather than arithmetic complexity.
In real-world physics applications, RMS velocity helps scientists analyze gas diffusion, molecular collisions, atmospheric science, and heat transfer mechanisms. For example, meteorologists use similar principles to study the movement of atmospheric gases, while chemical engineers apply these calculations when designing reactors, pipelines, and gas storage systems.
Another important concept linked to RMS velocity is the Maxwell-Boltzmann distribution, which describes how molecular speeds are distributed within a gas. According to this statistical model, not all molecules move at the same speed; instead, their velocities form a distribution curve. The RMS velocity lies slightly higher than the average molecular speed and provides a reliable measure of overall kinetic motion in the gas sample.
This Root Mean Square Velocity Calculator is especially useful for solving physics problems in topics such as:
• Kinetic Theory of Gases
• Thermodynamics
• Molecular Physics
• Chemical Engineering calculations
• Gas dynamics and diffusion
Students preparing for competitive exams such as JEE, NEET, IIT entrance exams, or university-level physics courses frequently encounter questions related to RMS velocity and gas molecular speeds. Using this calculator allows them to verify answers quickly and strengthen their understanding of how molecular motion depends on temperature and molar mass.
Another reason why RMS velocity is significant is that it connects microscopic molecular motion with macroscopic properties of gases, such as pressure and temperature. When molecules collide with the walls of a container, they exert force, which results in pressure. The faster the molecules move (higher RMS velocity), the greater the frequency and intensity of collisions, ultimately increasing the pressure of the gas system.
The RMS Velocity Calculator available on AllCalculators.co.in is designed to provide a fast, reliable, and user-friendly calculation experience for anyone studying physics or working with gas behavior. Whether you are solving homework problems, conducting laboratory experiments, or studying thermodynamic systems, this tool helps you determine molecular speed instantly and accurately.
By combining scientific accuracy, intuitive design, and real-time calculations, this physics calculator serves as an essential digital tool for students and professionals alike. Instead of struggling with complicated formulas, users can now calculate RMS velocity within seconds and gain deeper insights into the fascinating world of molecular motion.
🌍 Applications of RMS Velocity in Daily Life
Although RMS velocity is a theoretical concept in physics, it plays a major role in many real-world phenomena.
🌡️ Weather and Atmospheric Science: Meteorologists analyze the motion of atmospheric gases using principles similar to root mean square velocity. Faster molecular motion at higher temperatures influences wind movement, air pressure, and climate systems.
🧪 Chemical Engineering: Engineers designing gas reactors, pipelines, and pressure vessels rely on molecular speed calculations to ensure safe and efficient operation.
🚗 Combustion Engines: Gas molecules inside engines move rapidly during combustion. Understanding molecular motion helps engineers improve fuel efficiency and engine performance.
🧊 Refrigeration Systems: Cooling technologies depend on the behavior of gases under different temperatures. RMS velocity helps explain heat transfer and gas compression processes.
🧬 Scientific Research: Physicists and chemists studying molecular dynamics, diffusion of gases, and thermodynamic equilibrium frequently apply RMS velocity equations.
⚠️ Disclaimer
The RMS Velocity Calculator available on this website is designed for educational and informational purposes only. While every effort has been made to ensure accurate calculations using standard kinetic theory formulas and physics equations, results generated by this tool should be used as reference values and not as a substitute for professional scientific analysis or laboratory measurements.
Users are encouraged to verify calculations independently, especially when using results for academic research, engineering design, or professional applications. The creators of this tool are not responsible for any decisions or outcomes based on the calculated values.
📌 Related Calculator
❓FAQs
❓ What is RMS velocity in physics?
RMS velocity, or root mean square velocity, is the square root of the average of the squares of molecular speeds in a gas. It represents the effective average speed of gas molecules according to the kinetic theory of gases.
❓ What is the formula for RMS velocity?
The RMS velocity formula is: vᵣₘₛ = √(3RT / M)
Where R is the universal gas constant, T is temperature in Kelvin, and M is the molar mass of the gas.
❓ Why is RMS velocity important?
RMS velocity helps scientists understand molecular motion, gas pressure, temperature relationships, and thermodynamic behavior of gases.
❓ Does RMS velocity increase with temperature?
Yes. According to the formula, RMS velocity is proportional to the square root of temperature. Higher temperature means faster molecular motion.
❓ Which gas has the highest RMS velocity?
Gases with lower molar mass, such as hydrogen or helium, typically have higher RMS velocity compared to heavier gases at the same temperature.
