🔥 Latent Heat Calculator
Q = Heat Energy (J)
m = Mass (kg)
L = Latent Heat (J kg-1)
Table of Contents
✍️ Author & Academic Authority: Dr. Nitish Kr. Bharadwaj
📘 Qualifications: B.Sc., B.Ed., M.Sc., Ph.D. (Biochemistry), MBA (Financial Management)
🌡️ Latent Heat Calculator – Introduction
Instantly Calculate Heat Required for Phase Change ⚛️
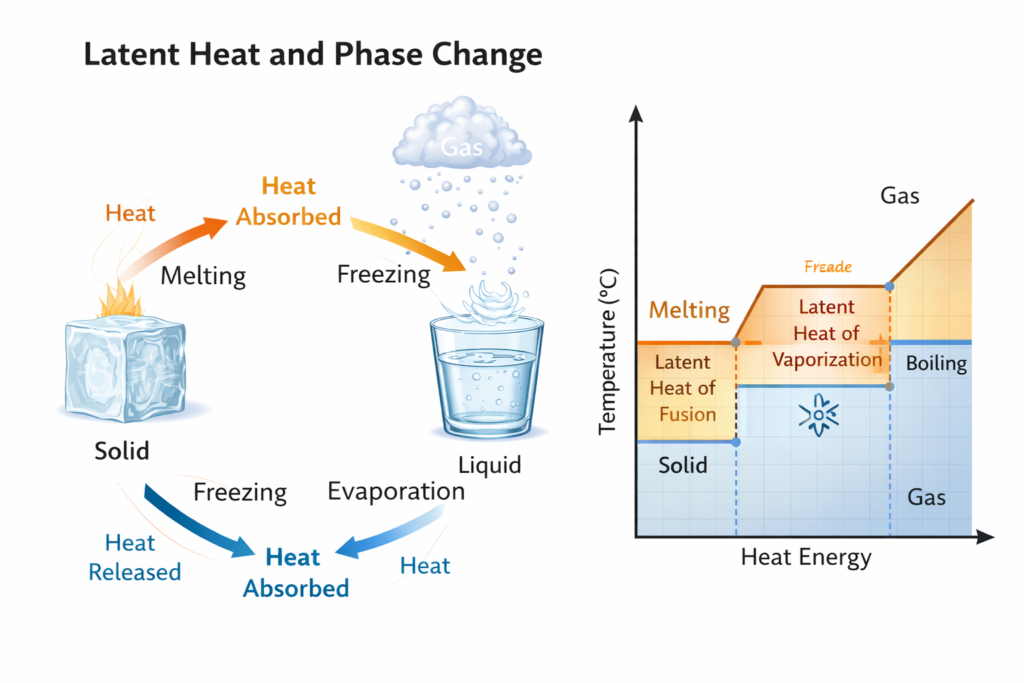
Understanding latent heat is fundamental in thermodynamics, physics, chemistry, and engineering. Whether water freezes into ice ❄️ or boils into steam 💨, heat energy is involved — but interestingly, the temperature remains constant during these phase changes. This hidden energy is called latent heat.
Our Latent Heat Calculator helps students, teachers, and engineers instantly determine the amount of heat energy required to change the phase of a substance without changing its temperature. Instead of performing complex manual calculations, you can simply enter the mass of the substance and its specific latent heat constant, and the calculator instantly determines the required energy using the latent heat formula.
The concept of latent heat was first introduced by the famous scientist Joseph Black. He discovered that when a substance changes state — such as from solid to liquid or liquid to gas — energy is absorbed or released without altering the temperature. This energy is essential to overcome the intermolecular forces that hold molecules together.
In physics, the latent heat formula is expressed as: Q = m × L
Where:
Q = Heat energy absorbed or released (Joules)
m = Mass of the substance (kg)
L = Specific latent heat (J/kg)
Using this formula, our online latent heat calculator determines the total energy involved in the phase change process.
For example, when ice melts into water, it absorbs energy known as the latent heat of fusion. Similarly, when water turns into steam during boiling, it absorbs the latent heat of vaporization. Even though heat energy is continuously added, the temperature remains constant until the entire phase change is completed.
This phenomenon occurs because the heat energy is not used to increase temperature. Instead, it is used to break intermolecular bonds between molecules.
The specific latent heat depends on the material. For instance:
Latent heat of fusion of ice ≈ 334,000 J/kg
Latent heat of vaporization of water ≈ 2,260,000 J/kg
By inputting these values into the latent heat formula calculator, students can quickly solve textbook problems, laboratory calculations, and real-world thermodynamic questions.
Our latent heat calculator tool is especially helpful for:
✔ Physics students solving thermodynamics problems
✔ Teachers explaining phase change concepts
✔ Engineers calculating heat transfer in industrial systems
✔ Chemistry learners studying energy transformations
The calculator eliminates calculation errors and provides instant results for heat energy calculations.
Another advantage of using an online physics latent heat calculator is speed and convenience. Instead of performing manual multiplication or searching for formula references, users can simply input values and obtain precise answers within seconds.
Latent heat plays a critical role in weather systems, refrigeration technology, steam engines, climate science, and industrial heat transfer processes. For instance, the evaporation of water from oceans absorbs large amounts of latent heat, which later releases during condensation to form clouds and storms.
In refrigeration systems, latent heat removal is used to cool environments efficiently. Refrigerants absorb latent heat when they evaporate and release it when they condense.
Similarly, in power plants, water converts into steam using latent heat energy, which then drives turbines to generate electricity.
By using our free online latent heat calculator, learners can better understand how heat energy behaves during phase transitions.
The calculator is designed with a simple user interface, ensuring that students can easily input data such as mass and latent heat constant without confusion. It is optimized for mobile devices, desktops, and tablets, making it convenient for quick problem solving anywhere.
If you’re studying thermodynamics, heat transfer, or phase change physics, this tool can dramatically simplify calculations and improve learning efficiency.
Try the Latent Heat Calculator now and instantly solve physics problems related to heat of fusion, heat of vaporization, and phase change energy.
🌍 Applications in Daily Life
Latent heat is not just a classroom concept. It plays an essential role in many real-world processes and technologies.
❄️ Ice Cooling Systems: When ice melts, it absorbs latent heat from surrounding objects, which helps keep beverages and food cold.
🌧️ Weather & Climate Systems: Water evaporation absorbs latent heat from oceans, while condensation releases it in clouds, influencing storms and rainfall.
🏭 Industrial Steam Production: Power plants use latent heat to convert water into steam that drives turbines for electricity generation.
🧊 Refrigerators & Air Conditioners: Cooling systems rely on latent heat absorption during refrigerant evaporation.
🍳 Cooking & Food Processing: Boiling water, steaming vegetables, and baking involve phase changes requiring latent heat.
🚰 Water Cycle: Evaporation, condensation, and freezing processes depend heavily on latent heat energy.
🏗️ Engineering & Thermodynamics: Engineers use latent heat calculations in heat exchangers, boilers, and cooling systems.
Using a latent heat calculator online makes solving such thermodynamic calculations quick and accurate.
⚠️ Disclaimer
The Latent Heat Calculator on allcalculators.co.in is designed for educational, academic, and general informational purposes only.
✔ The calculator provides estimated results based on the latent heat formula (Q = mL).
✔ Actual results may vary depending on material purity, environmental conditions, and measurement accuracy.
✔ Users should verify calculations when using results for scientific research, engineering applications, or professional analysis.
While we strive to ensure the highest level of accuracy and reliability, allcalculators.co.in cannot guarantee absolute precision of results. By using this tool, you acknowledge that the website is not responsible for decisions made based on calculator outputs.
📌 Related Calculator
❓FAQs
❓ What is latent heat in physics?
Latent heat is the amount of heat energy required to change the phase of a substance without changing its temperature. It occurs during processes such as melting, freezing, boiling, and condensation.
❓ What is the formula used in the latent heat calculator?
The latent heat formula is: Q = m × L
Where
Q = Heat energy (Joules)
m = Mass of the substance (kg)
L = Specific latent heat (J/kg)
❓ What are the types of latent heat?
There are mainly two types:
❄️ Latent Heat of Fusion – energy required to convert solid to liquid
💨 Latent Heat of Vaporization – energy required to convert liquid to gas
❓ Why does temperature remain constant during phase change?
During a phase change, the heat energy is used to break intermolecular forces between molecules instead of increasing temperature.
❓ Who discovered latent heat?
The concept of latent heat was discovered by Joseph Black, a Scottish physicist and chemist.
