⚛️ Bohr Model Calculator
Electron Orbit Radius:
rn = n2 × a0
Energy Level:
En = −13.6 / n2 (eV)
Table of Contents
✍️ Author & Academic Authority: Dr. Nitish Kr. Bharadwaj
📘 Qualifications: B.Sc., B.Ed., M.Sc., Ph.D. (Biochemistry), MBA (Financial Management)
⚛️ Bohr Model Calculator – Introduction
Instantly Calculate Atomic Energy Levels & Electron Orbits
The Bohr Model Calculator is an advanced physics tool that helps students, educators, and researchers quickly determine the energy level, orbital radius, and velocity of an electron in a hydrogen-like atom using the Bohr atomic model equations. This calculator simplifies complex atomic physics calculations by applying the principles developed by the Danish physicist Niels Bohr in 1913, making it easier to understand how electrons behave around atomic nuclei.
In atomic physics, the Bohr atomic model was a revolutionary concept that explained the structure of atoms by introducing quantized electron orbits. According to Bohr’s theory, electrons move around the nucleus in fixed circular paths called energy levels or shells. Each orbit corresponds to a specific energy value, and electrons can only jump between these levels by absorbing or emitting energy. This concept helped scientists explain the spectral lines of hydrogen, which classical physics failed to describe accurately.
Our Bohr Model Calculator uses the fundamental equations of the Bohr model to compute values such as the radius of the electron orbit (Bohr radius), electron energy level, electron velocity, and orbital frequency. By entering variables like the principal quantum number (n) and atomic number (Z), users can instantly determine important atomic properties without manually solving complex equations.
Understanding electron energy levels is crucial in fields like quantum mechanics, spectroscopy, atomic physics, and chemistry. For example, when an electron moves from a higher orbit to a lower orbit, it releases energy in the form of electromagnetic radiation. This process produces the characteristic spectral lines observed in hydrogen atoms. With the help of this Bohr orbit calculator, students can quickly analyze these transitions and understand the energy structure of atoms.
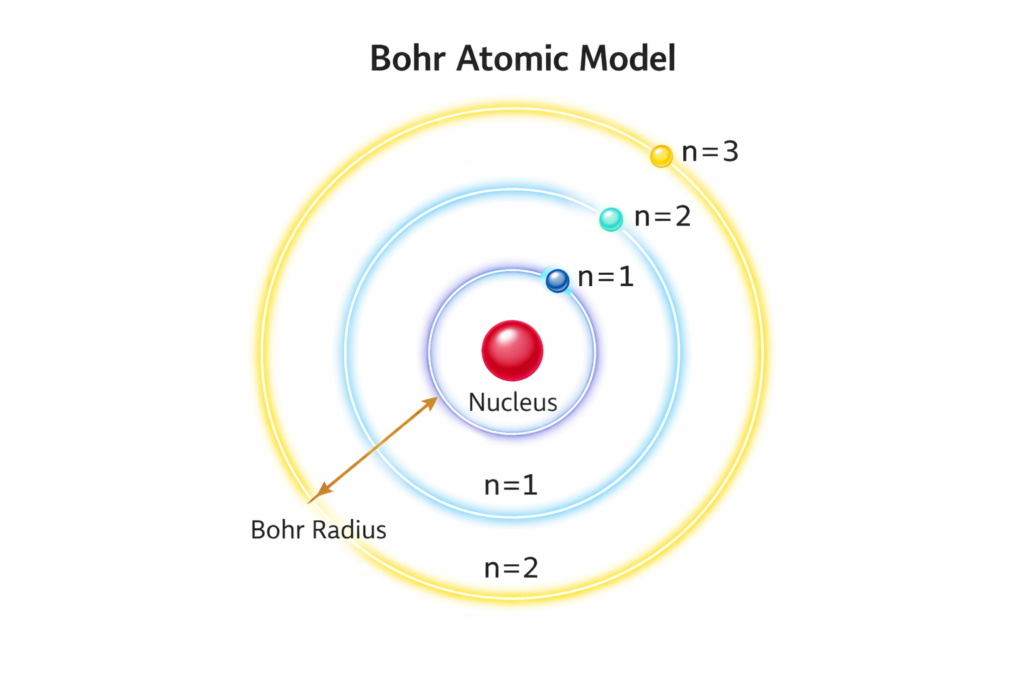
The Bohr model introduced several key formulas that remain foundational in modern physics education. One of the most famous equations describes the radius of the electron orbit, known as the Bohr radius equation: rₙ = n² × a₀ / Z
Where:
n = principal quantum number
a₀ = Bohr radius constant (5.29 × 10⁻¹¹ m)
Z = atomic number of the atom
Using this formula manually can be tedious, especially for large values of n. That’s why our Bohr radius calculator automates the calculation instantly and accurately.
Another important equation used in the Bohr model determines the energy of an electron in a particular orbit: Eₙ = −13.6 × Z² / n² (in electron volts)
This formula explains why electrons in lower orbits have more negative energy values, meaning they are more tightly bound to the nucleus. The Bohr energy level calculator on this page performs these calculations automatically, helping users understand the energy distribution within atoms.
Students studying quantum mechanics and atomic physics often struggle with complex calculations involving constants, units, and exponents. The Bohr atomic model calculator simplifies these calculations so users can focus on understanding the physical concepts rather than spending time on manual computation.
Another significant advantage of using this electron orbit calculator is accuracy. Manual calculations can easily lead to errors when dealing with scientific notation or constants such as Planck’s constant, Coulomb’s constant, or the electron mass. This calculator eliminates such mistakes and provides instant results.
Although the Bohr model works best for hydrogen and hydrogen-like atoms, it remains one of the most important stepping stones toward modern quantum mechanics. The model helped scientists understand atomic structure and paved the way for advanced theories like the Schrödinger wave equation and quantum orbital theory.
Today, the Bohr model is widely taught in schools, colleges, and physics courses because it provides a simple visual representation of atomic structure. The circular orbits introduced by Bohr make it easier for students to imagine how electrons move around a nucleus. Our Bohr Model Calculator enhances this learning process by providing real-time calculations that illustrate how changing quantum numbers affects atomic properties.
For example, increasing the principal quantum number increases the radius of the electron orbit and decreases the binding energy of the electron. This means electrons in higher energy levels are farther from the nucleus and require less energy to escape. Using the Bohr orbit radius calculator, users can observe these relationships instantly.
The tool is also helpful for solving physics homework problems, competitive exam questions, and engineering entrance exam practice. Many exams include questions related to the Bohr atomic model, hydrogen spectrum, and electron energy levels. Instead of spending several minutes solving each problem manually, students can verify their answers using this calculator.
Another important concept associated with the Bohr model is the quantization of angular momentum, which states that the angular momentum of an electron is restricted to discrete values: mvr = nħ
This principle laid the groundwork for modern quantum theory and remains an essential concept in physics education.
Our Bohr Model Calculator integrates these theoretical principles into a simple online interface. Users only need to enter the required parameters, and the calculator instantly displays the results. This makes it an ideal learning companion for students, teachers, researchers, and science enthusiasts.
Beyond education, atomic physics concepts also influence many modern technologies. For instance, laser technology, atomic clocks, spectroscopy, and semiconductor devices all rely on the behavior of electrons in atoms. Understanding the Bohr energy level equation helps explain how these technologies work at the microscopic level.
The calculator on this page is designed to be fast, mobile-friendly, and highly accurate, allowing users to perform calculations anytime and anywhere. Whether you are studying physics, preparing for exams, or exploring atomic theory out of curiosity, this Bohr Model Calculator provides a reliable and easy way to analyze atomic properties.
By combining scientific formulas, intuitive design, and instant computation, this tool transforms complicated physics equations into simple calculations that anyone can understand.
🧠 Applications in Daily Life ⚛️
Although the Bohr atomic model is mainly used in physics and chemistry, its concepts influence many technologies used in daily life.
🔬 Spectroscopy and Chemical Analysis: Scientists use electron energy transitions predicted by the Bohr model equations to identify elements in laboratories and astronomical observations.
💡 Laser Technology: Lasers work based on electron transitions between energy levels. The Bohr energy level concept helps explain how photons are emitted when electrons drop to lower orbits.
🛰 Atomic Clocks: Atomic clocks rely on electron transitions in atoms such as cesium. These clocks provide extremely precise time measurement used in GPS systems and satellite navigation.
📡 Telecommunication Technology: Understanding atomic energy levels helps design semiconductors, LEDs, and photonic devices used in modern communication systems.
🌌 Astrophysics and Space Research: Astronomers analyze spectral lines from stars and galaxies using the Bohr atomic model theory to determine the chemical composition of distant celestial bodies.
📚 Education and Physics Learning: Students worldwide use Bohr model calculators to learn atomic structure, solve physics problems, and understand quantum mechanics concepts.
⚠️ Disclaimer
The Bohr Model Calculator available on this website is designed for educational and informational purposes only. While the formulas implemented in this calculator are based on standard physics equations from the Bohr atomic model, results should be used as a reference for learning, homework verification, and conceptual understanding.
This tool primarily applies to hydrogen and hydrogen-like atoms, where the Bohr model provides accurate predictions. For multi-electron atoms or advanced quantum mechanical calculations, more sophisticated models such as quantum orbital theory and Schrödinger equations are required.
Although we strive to maintain high accuracy and reliability, users should always verify results when using calculations for academic exams, research, or professional work.
By using this calculator, you agree that allcalculators.co.in is not responsible for any decisions made based on the results provided by this tool.
📌 Related Calculator
❓ FAQs
❓ What is the Bohr Model Calculator?
The Bohr Model Calculator is an online physics tool that calculates the electron orbit radius, energy level, and velocity of an electron in a hydrogen-like atom using the Bohr atomic model equations.
❓ What is the Bohr radius?
The Bohr radius is the average distance between the nucleus and the electron in the ground state of a hydrogen atom. Its value is approximately 5.29 × 10⁻¹¹ meters.
❓ What is the formula for the Bohr model energy level?
The Bohr energy equation is: Eₙ = −13.6 × Z² / n² (eV). where Z is the atomic number and n is the principal quantum number.
❓ For which atoms is the Bohr model accurate?
The Bohr model works best for hydrogen and hydrogen-like atoms such as He⁺ and Li²⁺ because they contain only one electron.
❓ Why is the Bohr model important in physics?
The Bohr atomic model introduced the concept of quantized energy levels, which became the foundation for modern quantum mechanics and atomic physics.
